Description of Lab Projects
Cellular responses to activation of growth factor receptors is dictated by the repertoire of signaling molecules present within a given cell type. We identified a novel mechanism utilized by the CSF-1R for activating the phosphatidylinositol 3-kinase (PI3K)/Akt survival pathway in the 32D myeloid cell line (Fig. 1). Unlike the previously described pathway involving direct CSF-1R:PI3K binding, this novel mechanism is activated by a deletion mutant of CSF-1R (DKI) that cannot bind PI3K. Using both pharmacological and genetic approaches, we determined that the novel pathway involves a CSF-1R-Src-Gab2-PI3K link. Src family kinases are cytosolic tyrosine kinases whereas Gab2 is a recently identified docking/scaffolding molecule that may play a particularly important role in hematopoietic systems. Current research focus is to further delineate the roles of Src and Gab2 in mediating CSF-1 signals necessary for growth and survival.
Additional recent studies from our lab have revealed that the DKI mutant is mitogenically defective and undergoes a novel form of cell death that appears to be largely independent of caspases and mitochondrial dysfunction. Complementation with constitutively active downstream signaling molecules in combination with gene expression microarray profiling are in progress to further elucidate the mechanisms of cell growth and death.
Recent exciting results in the lab point to an essential role for Gab2 in maintaining neural stem cell proliferation (self-renewal). Since a multitude of receptor tyrosine kinases are found in neural stem cells and their differentiated progeny (neurons, astrocytes and oligodendrocytes), this has significant implication.
In addition, we have turned to mouse models to examine how CSF-1 and CSF-1R regulate the commitment of hematopoietic progenitors to the monocyte/macrophage lineage. As monocytes and macrophages are major effectors of innate immunity and integral modulatory cells found in many pathological conditions such as cancer, diabetes and Alzheimer's disease, it is important to understand how these cells are recruited from the bone marrow. A future goal is to understand the origin of tumor associated macrophages (TAMs) and the signals that promote their recruitment to tumor sites.
Further description of individual projects is provided in the individual links.
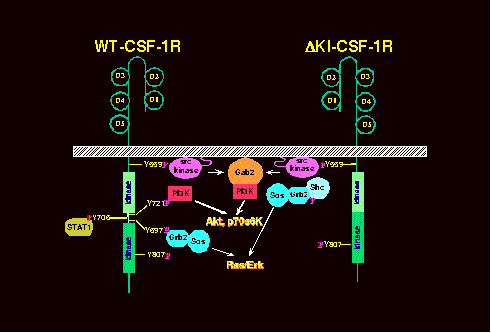
Lee and States, MCB 20:6779,2000
Fig. 1